Zynamite has been studied in multiple clinical trials in mental energy, cognitive and physical performance settings.
In the most recent clinical study, 88 healthy university students were given a single dose of 100 mg Zynamite S or a matching placebo and then assessed three times over a 5-hour period. The study featured a randomized, double-blind, placebo-controlled crossover design with a 7-day washout between testing conditions. Assessments were performed at baseline, 30 minutes, 3 hours, and 5 hours.
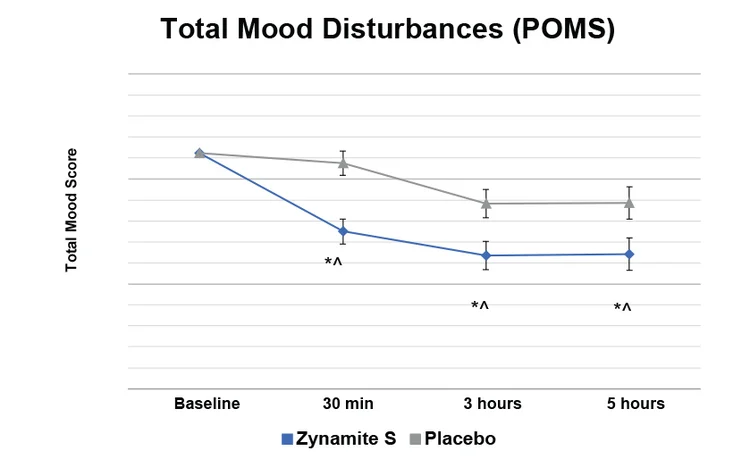
Zynamite subjects experienced improved mood starting at 30 minutes, with a 24.1% improvement in 5 hours in Total Mood Disturbances on the Profile of Mood States (POMS) tests.
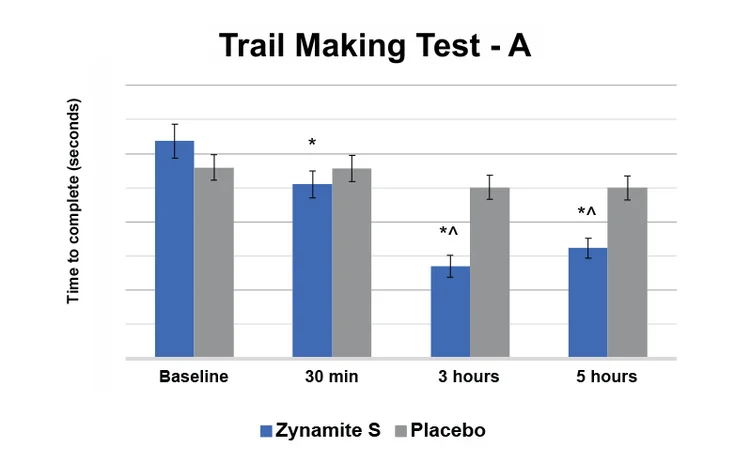
An improvement of 25.4% in processing speed was demonstrated by performance on Trail Making Test A
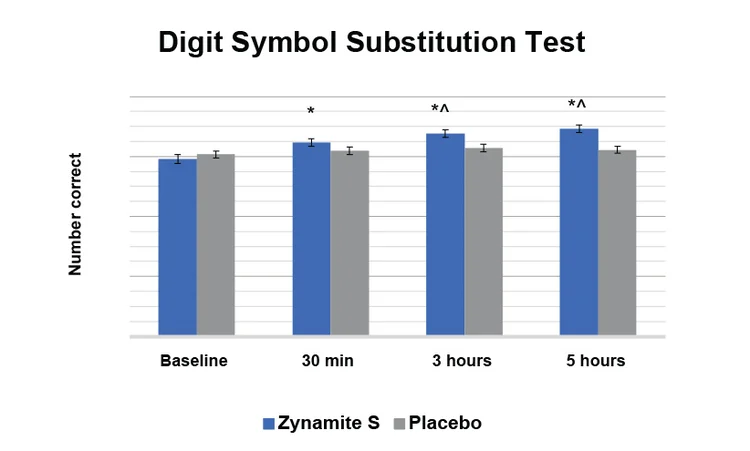
Subjects taking Zynamite S improved their performance on the Digit Substitution Test by 17% at 5 hours, with significant differences vs. placebo at 3 hours
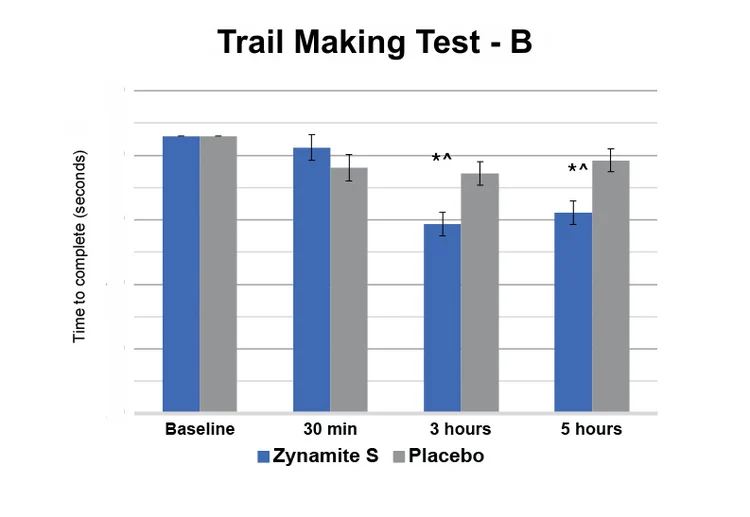
Subjects saw a greater than 21.1% improvement in the ability to handle complex tasks, as demonstrated by performance on Trail Making Test B differences vs. placebo at 3 hours
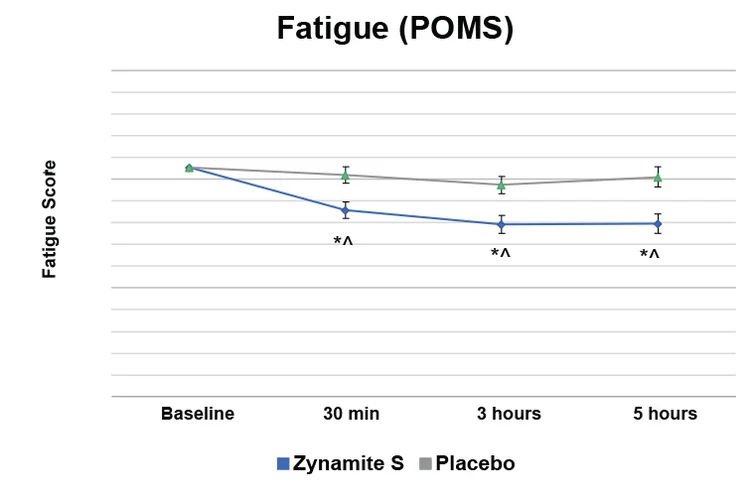
Subjects taking Zynamite S experienced 26% less fatigue at the earliest measurement point
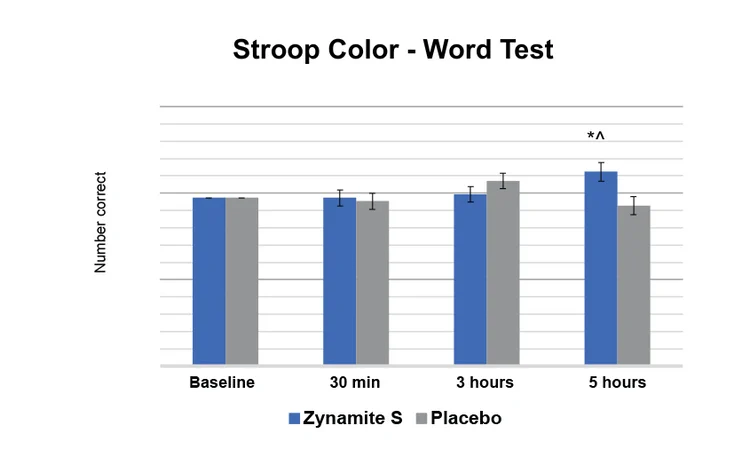
Subjects taking Zynamite S improved 4.7% on the Stroop Color-Word Test
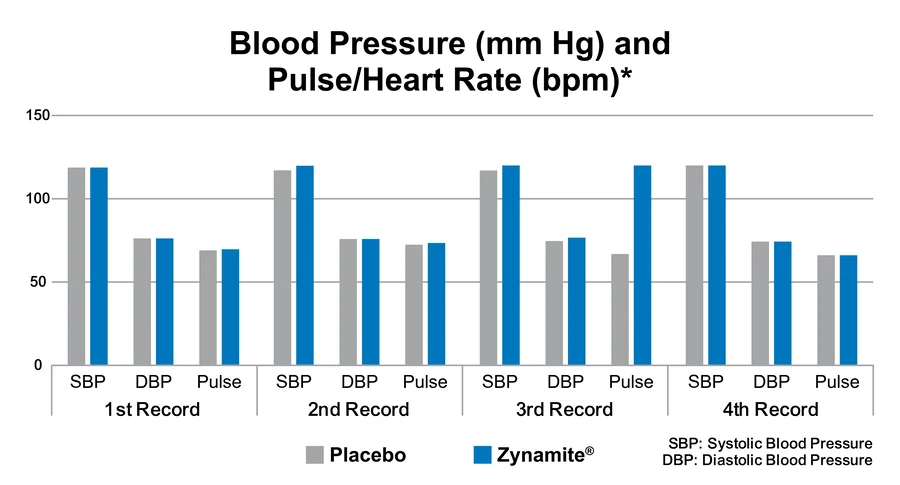
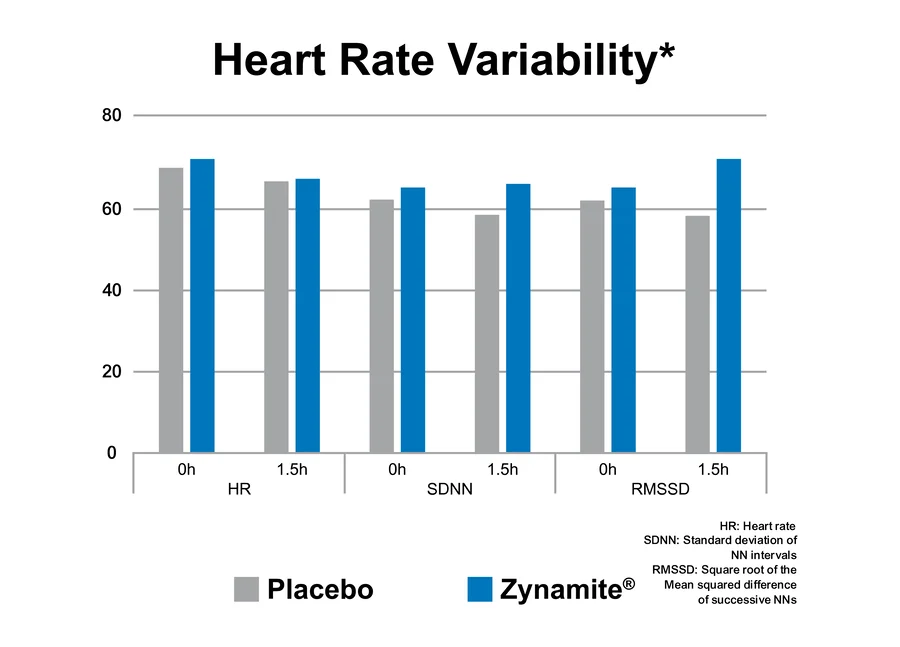
Zynamite is described as a ‘non-stim’ ingredient. A clinical study published in the Journal of Ethnopharmacology found that while Zynamite and its combination with other ingredients, e.g., tigernut (chufa) had caffeine-like effects on performance of cognitively challenging tasks, it did not produce any changes in blood pressure, pulse, or heart rate variability.